Physics
Fox
Summary
- Anything that takes up space is known as matter.
- Matter can be in 3 states: solid, liquid and gas.
- All matter is made up of tiny spherical objects called atoms (even you!) The existence of atoms explains why matter has 3 states:
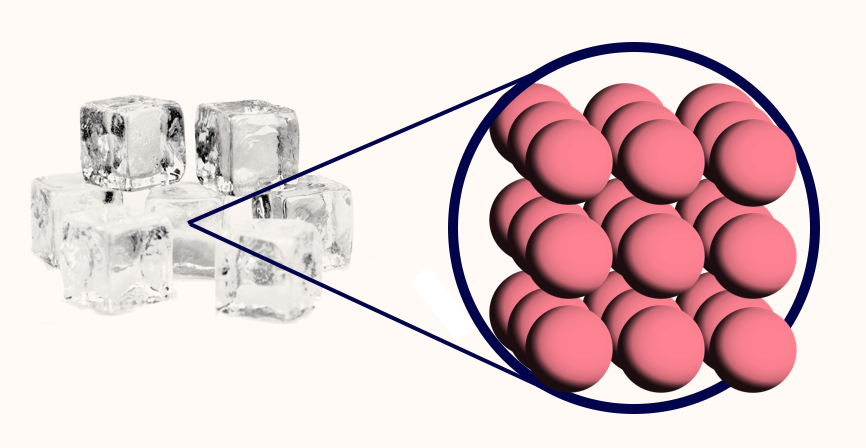
- Solid: Atoms are together in a lattice.
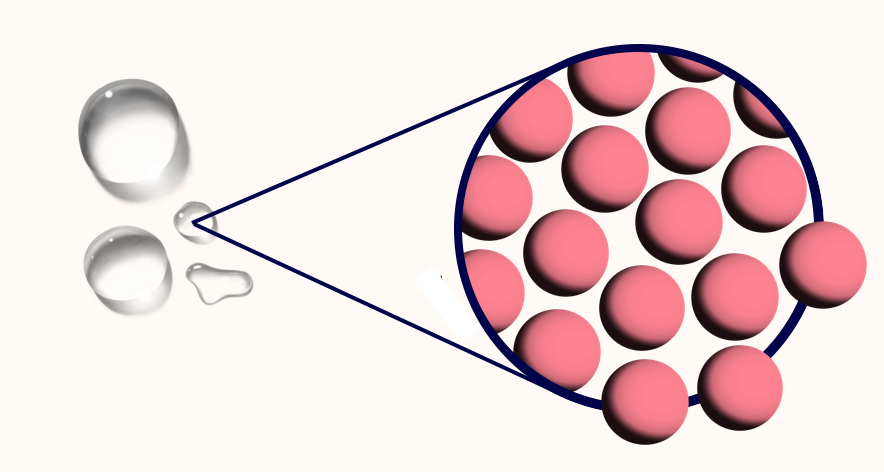
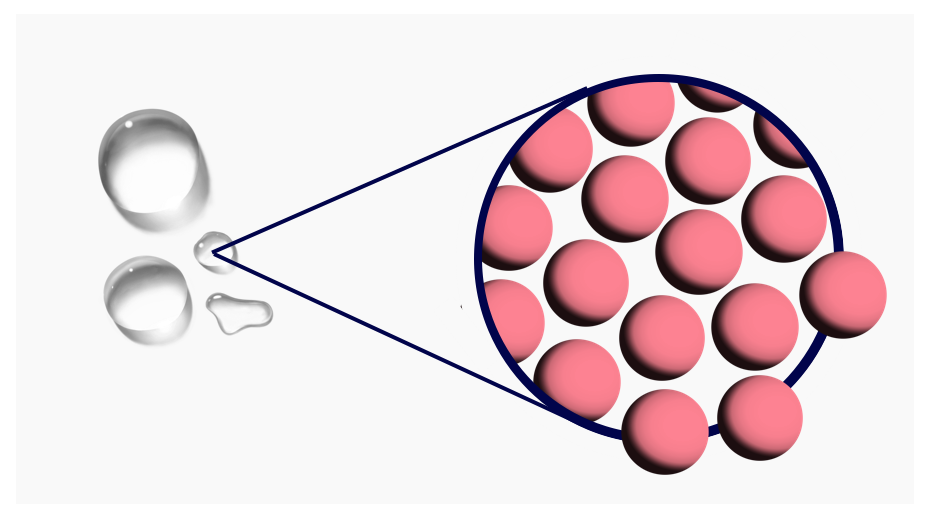
- Liquid: Atoms are together, but can slide past each other.
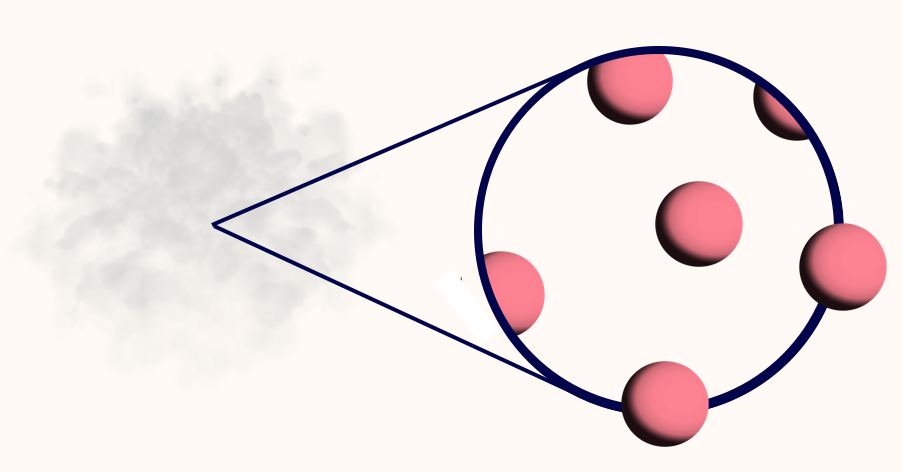
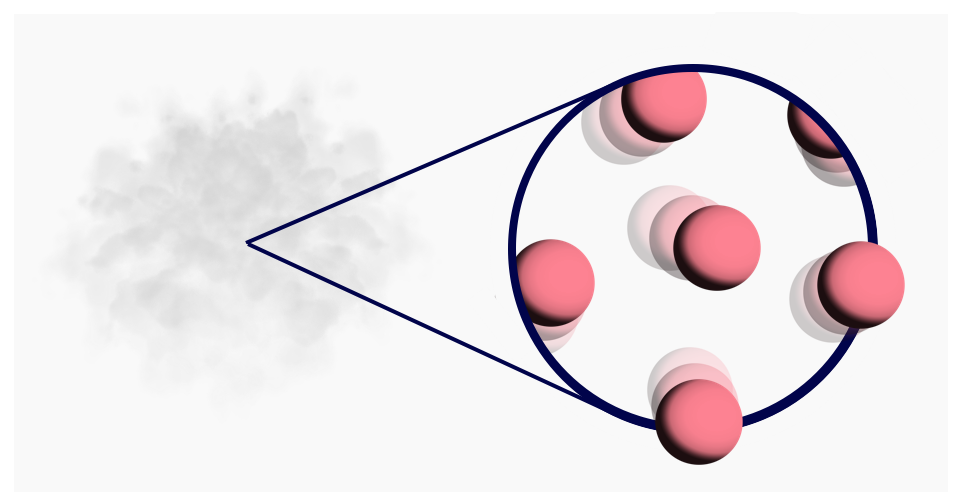
- Gas: Atoms are separated from each other.

Everything you can see around you is called matter; furniture, plants, elephants, the moon, whatever.

Even some things you can't see (like air) also count as matter. Matter is really anything that takes up space.
All matter exists in one of three types (called states): Solid, liquid and gas. We can see all three in this cup of tea:
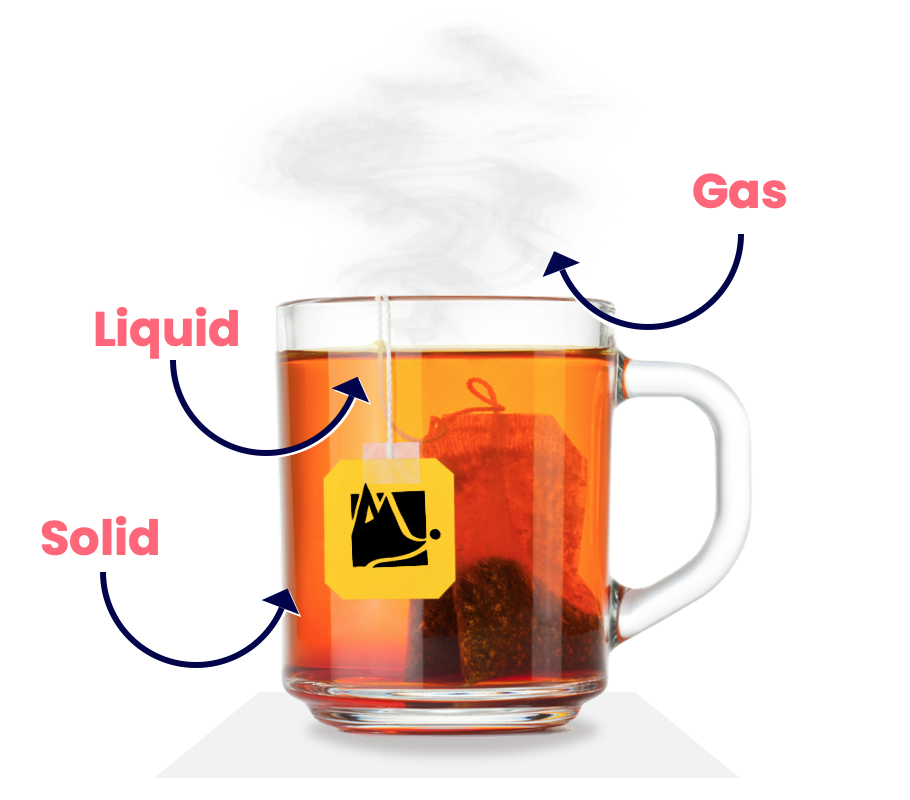
Matter is able to change state; if an ice lolly (a solid) gets too hot, it will turn into juice (a liquid) and drip off the stick — a mournful experience.

Why does matter have these 3 states? And how come it can change between them? The answer: atoms.
All matter is made of atoms.
Atoms are tiny spherical objects. They are all slightly attracted to each another, so if you have enough atoms (and it's cold enough) they will clump together into a grid known as a lattice. This is what a solid is.
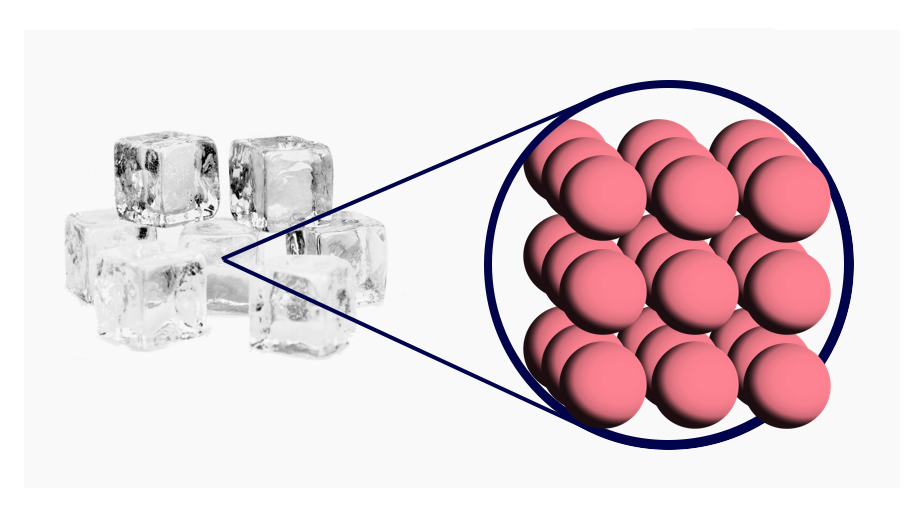
This lattice can be seen with special type of microscope; here's a real image of zoomed-in solid gold:

Image credit: Ye et al
It's important to stress how absolutely tiny atoms are. A common analogy is that there are more atoms in a grain of sand then there are grains of sand on all the Earth's beaches.
Here's another way to look at it: A kilogram bar of gold costs £42,000. But it takes so many atoms to make this bar that a single gold atom is worth only 0.000000000000000001p. Even one play on Spotify is worth more than that.

The atoms in a lattice are never sitting still; they're always jiggling in place a little bit. The more you heat up a material, the more its atoms shake around.

At a hot enough temperature, the atoms are jiggling so much that they can no longer stay in a lattice structure — now they slide past each other. The state of the material has changed; it is now a liquid.
At even hotter temperatures, the atoms are jiggling far too quickly to remain neighbours — the atoms separate from each other. The material is now in the third and final state: a gas.
On the next page, we'll learn more about the change of state between solids, liquids and gases.
As tiny as they are, atoms are themselves made of even smaller particles. We'll look into that later!
Congratulations!
9 of 9 questions completed
+ ⭐️ collected.
Sign up (for free!) to:
• save your progress 📊
• create constellations✨
• customise your fox! 🦊







